Balance on the edge of ions
Kidney disease foreshadows changes in the blood
An ongoing, silent battle for balance is waged in our bodies—not of the soul, but of chemistry. Research conducted by scientists from Wroclaw Medical University reveals that a simple ratio—the relationship between two molecules, chloride and bicarbonate—can signal when a patient with kidney disease may need to begin dialysis. Once again, medicine shows that the simplest solutions can be the most powerful.
The biochemistry stage
You breathe, eat, and run to catch a tram. Every one of these actions triggers chemical reactions in your body. Thousands of molecules work within fractions of a second to restore balance. Among the key players in this biochemical performance are your kidneys.
Their job is not just to filter waste products from the blood. They are something much more: internal chemists, guardians of balance. They determine how much acid and how much base remain in the bloodstream. Their vigilance ensures that our internal pH—the measure of acidity—neither drops too low nor climbs too high. In this game, there is no room for error.
Balance on the edge of ions: Podcast
Click here to listen!
The silent opponent
In metabolic acidosis, the body loses its delicate balance. The blood becomes too acidic, meaning its pH falls below the physiological level. A low pH disrupts equilibrium and affects various bodily functions, including muscle function, heart activity, and cognitive performance.
Acidosis often develops slowly and quietly, without alarming symptoms. That's why in medicine, it's sometimes called the "silent epidemic" among patients with chronic kidney disease (CKD). In these patients, the buffer system begins to fail, and the kidneys can no longer keep up with acid removal.
"In advanced CKD, we observe a significant drop in blood bicarbonate levels and a simultaneous rise in chloride concentration. This shift in ion ratios is not coincidental—it reflects impaired renal excretory function," says Dr. Tomasz Gołębiowski from the Department of Nephrology, Transplantation Medicine, and Internal Medicine at Wroclaw Medical University.
The pH scale ranges from 0 to 14—the lower the number, the more acidic the solution. Human blood must remain within a very narrow range: 7.35–7.45, about the same as seawater. When pH falls below 7.35, acidosis begins. A pH of 7.33 or lower indicates advanced metabolic acidosis.
Salt vs. Soda – a chemical scale
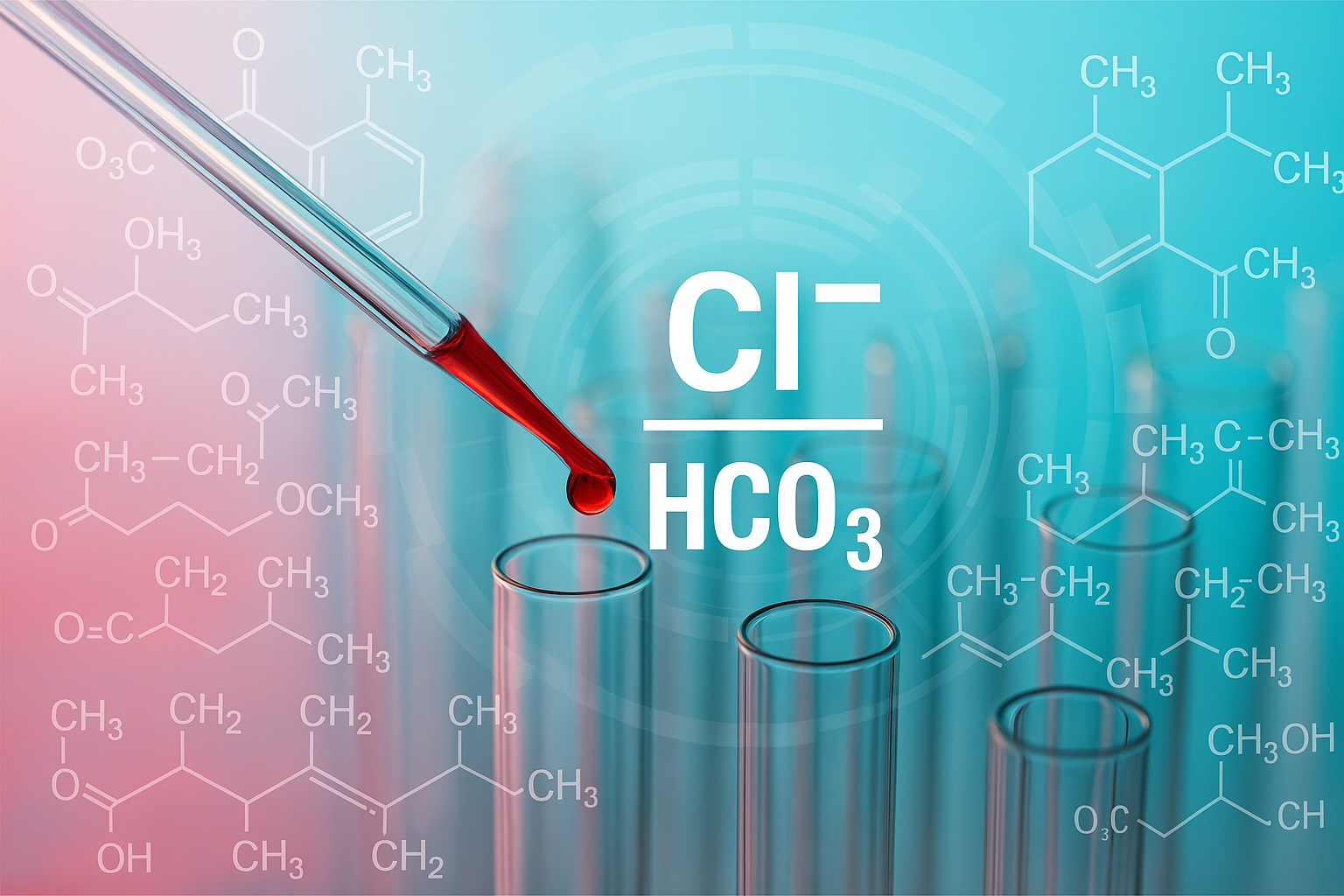
Researchers at Wroclaw Medical University decided to examine acidosis from a different perspective. Instead of solely measuring bicarbonate (HCO₃⁻) levels, they proposed a new indicator: the Cl⁻/HCO₃⁻ ratio, which compares chloride to bicarbonate.
Think of it as a seesaw between two anions with opposing roles in the chemical system. Bicarbonates neutralize acids. Chlorides, although necessary, tend to accumulate when the kidneys are not functioning correctly. As one anion rises and the other falls, the body's chemical balance begins to teeter. And this ratio reveals more than the current diagnostic gold standard.
"The Cl⁻/HCO₃⁻ ratio can not only confirm the presence of acidosis, but also indicate its severity and even estimate the time to dialysis initiation," adds Dr. Gołębiowski.
A simple ratio
The study involved 115 patients with advanced kidney disease. Blood samples were taken during the creation of an arteriovenous fistula for dialysis. Researchers measured chloride and bicarbonate concentrations.
The results were surprisingly precise. When the Cl⁻/HCO₃⁻ ratio exceeds 6.22, there is an 87% probability that the patient already has advanced metabolic acidosis. Moreover, these patients began dialysis an average of 45 days earlier than others.
The indicator proved more accurate than bicarbonate concentration alone and was superior to the classic anion gap, a measure used to detect hidden toxins.
"The strength of our approach lies in its simplicity: Cl⁻ and HCO₃⁻ are routinely measured in blood tests. Their relationship can provide physicians with an additional tool in everyday clinical practice," summarizes the Wroclaw nephrologist.
Rethinking thresholds
For years, the threshold for treating metabolic acidosis was a bicarbonate level below 22 mmol/L, prompting the use of sodium bicarbonate supplementation. However, recent international guidelines have lowered this threshold to 18 mmol/L due to insufficient evidence of clear benefit from supplementation at higher levels. This shift reflects the ongoing uncertainty about the optimal treatment threshold. Guideline authors have called for more research, noting that the ineffectiveness of bicarbonate therapy in some patients may stem from a more complex disorder than previously understood.
It may not be just about adjusting numbers, but also changing the qualitative approach. Instead of focusing on a single parameter, it may be time to examine the interplay between molecules: the buffer (bicarbonate) and its counterforce (chloride). Just as financial analysts examine both income and expenses, physicians could consider the Cl⁻/HCO₃⁻ ratio—a chemical balance sheet.
Primarily, this indicator serves as a forecast, as it not only reflects the current state but also warns of potential developments in the weeks ahead.
For now, Cl⁻/HCO₃⁻ remains a prognostic tool requiring further study. But if its effectiveness is confirmed in broader patient groups, it could become not only a new diagnostic standard, but also a guide for personalized treatment and reduced use of unnecessary medications.

An early warning?
Although the study focused on patients with advanced kidney disease, the authors suggest the indicator should be evaluated earlier, particularly at stage G3 of CKD. Why then? Because this is when "subclinical acidosis" often appears: a state in which serum bicarbonate levels may still fall within lab norms, but the body is already beginning to suffer from acid buildup.
During this stage, excess acid is buffered at the expense of bones and soft tissues. The function of the renal tubules—which is responsible for excreting ammonium and phosphate—is already impaired. Standard parameters show no red flags, but tissue damage is quietly progressing.
It's in these silent, "transitional" stages that the Cl⁻/HCO₃⁻ ratio may prove especially valuable. It captures two simultaneous shifts: a declining bicarbonate level and a compensatory rise in chloride, sending a clearer warning signal than either parameter alone.
"We believe expanding the study to include G3-stage patients could help identify those at higher risk of rapid disease progression, due to more advanced tubular dysfunction," says Dr. Gołębiowski.
This, in turn, could allow for gentler but earlier interventions —such as dietary, pharmacological, or microbiome-targeted—before irreversible damage occurs. Shifting from "firefighting" to "detecting smoke" early could be crucial for slowing disease progression.
What if the solution lies in the kitchen?
Over the years, various methods of treating acidosis have been tried. Oral bicarbonates? Sometimes effective—but in high doses, they can raise blood pressure and worsen heart failure symptoms, especially in patients with cardiovascular disease.
A new drug, veverimer, was once considered a breakthrough—safer and sodium-free. However, recent studies have tempered that optimism: veverimer did not slow kidney function decline, although research is ongoing.
In the background looms one persistent question: Can we influence acid–base balance through diet? Growing evidence suggests we can. Some studies indicate that a diet rich in vegetables, fruits, and legumes—naturally alkaline—can improve blood acidity parameters as effectively as bicarbonate supplementation.
Even better, such diets offer additional benefits, including lower blood pressure, improved lipid profiles, and better glycemic control.
This represents a new model of care—less invasive, more personalized. Perhaps what we eat matters more than we think. Maybe acid–base balance is not just a medical issue—it's a lifestyle.
And new indicators like Cl⁻/HCO₃⁻ may not be a verdict, but a warning—a signal that it's time to slow down, change our menus. After all, the most effective remedy for biochemical imbalance may already be in our grocery basket.
D. Sikora

FAQ: Balance on the edge of ions
What is the main finding of the study on metabolic acidosis in patients with PChN?
The primary finding of the study is that the chloride-to-bicarbonate ratio (Cl/HCO₃) may be a strong predictor of advanced metabolic acidosis in patients with chronic kidney disease (CKD) at stages G4 and G5. In particular, a Cl-/HCO₃- ratio higher than 6.22 (mmol/l)/(mmol/l) indicates advanced metabolic acidosis. This ratio also showed higher predictive power than bicarbonate concentration alone.
How do chloride and bicarbonate levels change with the progression of PChN, and why is their ratio important?
As glomerular filtration rate (eGFR) decreases in PChN, bicarbonate (HCO₃-) levels decrease because they are used to buffer accumulating endogenous acids. At the same time, chloride (Cl-) concentration increases to compensate for changes in the anion gap. The study showed a strong negative correlation between Cl- and HCO₃- concentrations. The Cl-/HCO₃- ratio combines these two key ions into a single parameter, reflecting both the severity of acidosis (low HCO₃-) and compensatory mechanisms or accumulation of other anions (high Cl-). This composite index offers a more comprehensive assessment of acid-base imbalance than examining each parameter separately.
How does the Cl-/HCO₃- ratio compare with other known acid-base balance indicators in predicting advanced acidosis?
The Cl-/HCO₃- index showed high resolving power in predicting advanced acidosis (pH ≤ 7.33), reaching an AUC (area under the ROC curve) of 0.922. This result was comparable to Base Excess (BE-A, AUC = 0.956) and bicarbonate (HCO₃--A, AUC = 0.911), and better than for chloride (Cl--A, AUC = 0.903), eGFR (AUC = 0.584), and creatinine (AUC = 0.626) levels alone. Notably, the arterial blood anion gap (AG-A) was not a reliable predictor of arterial pH (AUC = 0.589), underscoring the distinct prognostic value of the Cl-/HCO₃- ratio.
What are the current recommendations for the treatment of metabolic acidosis in PChN, and what controversies surround them?
The 2012 KDIGO guidelines recommended diagnosing metabolic acidosis at bicarbonate levels below 22 mmol/L and suggested drug treatment. However, more recent studies, including BiCARB, have questioned the clear benefits of oral bicarbonate supplementation, showing, among other things, an increased risk of heart failure when levels are too high (above 24-26 mmol/L). Accordingly, the new 2024 KDIGO recommendations have lowered the intervention threshold to 18 mmol/L, emphasizing not the aggressive normalization of levels, but the prevention of severe acidosis. This shift in emphasis promotes the search for alternative or additional indicators to help guide therapy on an individual basis.
Why is the new metabolic acidosis index in patients with MS important?
Metabolic acidosis is a common and serious complication in patients with PChN. Although bicarbonate levels have traditionally been used to diagnose and treat it, there is controversy over optimal supplementation, as some studies suggest potential risks associated with raising these levels too aggressively. Other indicators, such as the anion gap, are susceptible to the influence of various factors and do not always correlate well with pH. Therefore, a more precise and reliable indicator, such as Cl/HCO₃, can better assess the severity of acidosis and help inform personalized therapeutic decisions.
What is the connection between the Cl-/HCO₃- ratio and the need to start dialysis therapy?
The study showed that patients with a higher Cl-/HCO₃- ratio (≥ 6.22 (mmol/l)/(mmol/l)) started dialysis significantly earlier than those with a lower ratio. The median time to dialysis initiation was 49 days in the high-index group, compared to 94 days in the low-index group. This suggests that a higher Cl-/HCO₃ ratio may indicate more advanced kidney disease and faster progression to renal replacement therapy.
How does “metabolic acidosis with normal anion gap (AGMA)” differ from “acidosis with increased anion gap (HAGMA)” in patients with PChN?
There are two main types of metabolic acidosis in PChN. AGMA (normal anion gap) is typical of the early stages of the disease, where bicarbonate levels drop, but the anion gap remains normal due to a compensatory increase in chloride. HAGMA (elevated anion gap) occurs in later stages (G4 and G5), when the kidneys are significantly damaged and unable to remove inorganic anions (e.g., phosphate, sulfate) and organic anions (e.g., indoxyl, p-cresol), leading to an increase in the anion gap. The study showed that patients with HAGMA had lower eGFR and started dialysis faster compared to patients with AGMA.
What are the study's limitations and directions for future research?
The study was cross-sectional, which limits the ability to track changes in POCT parameters over time; however, a two-year follow-up of dialysis initiation provided valuable long-term data. The study group included only patients with PChN in stages G4 and G5, which limits the generalizability of the results to earlier stages. Urinary parameters such as ammonia excretion or titratable acids were also not analyzed, nor was the effect of diet considered. Arterial blood was used, which may differ from the more commonly used venous blood in practice. Despite these limitations, the study provides a strong basis for further analysis, suggesting that the Cl-/HCO₃ ratio may become a valuable tool for individualizing the treatment of patients with metabolic acidosis and PChN.
FAQ by Notebook LM

This material is based on an article:
The ratio of chloride to bicarbonate is a predictor of advanced metabolic acidosis in CKD stages G4 and G5
Justyna Korus, Maciej Gołębiowski, Jakub Stojanowski, Maciej Szymczak, Marcelina Żabińska, Dorota Bartoszek, Katarzyna Kościelska-Kasprzak, Mirosław Banasik, Milena Ściskalska, Mariusz Kusztal, Tomasz Gołębiowski
Scientific Reports
DOI: 10.1038/s41598-025-05633-6
Web. A. Hasiak